A copper disc perforated with numerous holes was placed across the cylinder recessed down from the top. His original design consisted of a 3.5 inch diameter copper cylinder. In this design the original perforated disc has become a cylinder inside the upper part of the cell to hold copper sulfate crystalsĭaniell first constructed his cell in 1836. The Daniell cell can be used to ‘generate’ electricity, by consuming an electrode, or to store electricity.ĭevelopment Daniell's original construction ĭiagram of early Daniell cell published by Daniell in 1839. Hence, the Daniell cell is reversible, if the current drawn from (or fed to) it is small.

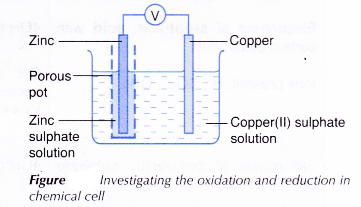
a battery charger) such that the potential difference of the source is slightly higher than the cell emf (1.1 v) then the current flow could be reversed and the reaction would become: If the cell is connected to a potential source (e.g. At the same time, potassium ions from the salt bridge move into the copper half-cell in order to replace the Cu 2+ ions being precipitated onto the copper electrode. In the above wet-cell during discharge, nitrate anions in the salt bridge move into the zinc half-cell in order to balance the increase in Zn 2+ ions. The salt bridge typically contains a high concentration of potassium nitrate (a salt that will not interfere chemically with the reaction in either half-cell). When the half cells are placed in two entirely different and separate containers, a salt bridge is often used to connect the two cells. A porous barrier or ceramic disk may be used to separate the two solutions while allowing the flow of sulfate ions. Since neither half reaction will occur independently of the other, the two half cells must be connected in a way that will allow ions to move freely between them. This provides an electrical current that illuminates the bulb. Excess electrons produced by the oxidation of zinc metal are “pushed” out of the anode, which is therefore the negative electrode, travel through the wire and are "pulled" into the copper cathode where they are consumed by the reduction of copper ions. A wire and light bulb may connect the two electrodes. The two half cells each support one half of the reactions described above. In classroom demonstrations, a form of the Daniell cell known as two half cells is often used due to its simplicity. This energy can mostly be attributed to the 207 kJ/mol weaker bonding (smaller magnitude of the cohesive energy) in zinc compared to copper metal, which can be explained in terms of the lack of bonding via partially filled d-orbitals in zinc. The Daniell cell produces approximately 213 kJ per mole (65 g) of zinc. These processes result in the accumulation of solid copper at the cathode and the corrosion of the zinc electrode into the solution as zinc cations. Note that positively charged copper ions move towards the positive electrode, driven by a reduction in chemical energy. ( Standard electrode reduction potential +0.340 V ) ( Standard electrode reduction potential −0.7618 V ) Īt the cathode (positive electrode), copper is reduced as per the following reaction:Ĭu 2+ (aq) + 2e − → Cu (s). The two-half-cell form for classroom demonstrations Zn (s) → Zn 2+ (aq) + 2e −.

With contemporary definitions, the standard potential of the Daniell cell at 25 ☌ is actually 1.10 V. The definitions of electrical units that were proposed at the 1881 International Conference of Electricians were designed so that the electromotive force of the Daniell cell would be about 1.0 volts.

The Daniell cell is also the historical basis for the contemporary definition of the volt, which is the unit of electromotive force in the International System of Units. A later variant of the Daniell cell called the gravity cell or crowfoot cell was invented in the 1860s by a Frenchman named Callaud and became a popular choice for electrical telegraphy. The Daniell cell was a great improvement over the existing technology used in the early days of battery development. Zinc sulfate may be substituted for the sulfuric acid. He was searching for a way to eliminate the hydrogen bubble problem found in the voltaic pile, and his solution was to use a second electrolyte to consume the hydrogen produced by the first. The Daniell cell is a type of electrochemical cell invented in 1836 by John Frederic Daniell, a British chemist and meteorologist, and consists of a copper pot filled with a copper (II) sulfate solution, in which is immersed an unglazed earthenware container filled with sulfuric acid and a zinc electrode.

